News
Pediatric Low-Dose Patch Approved! Tulobuterol Patches (Deruituo®) Carefully Protects Respiratory Health for All Ages
Release Date: 2026-01-28
On January 26, the 0.5 mg/patch pediatric low-dose formulation of the Tulobuterol Patches (Deruituo®), the first domestic generic developed by Beijing Tide Pharmaceutical, a subsidiary of Sino Biopharm (1177.HK), officially received a drug registration certificate from the National Medical Products Administration (NMPA). Following the approval and launch of the 2 mg/patch adult formulation, this marks a further refinement of Deruituo® for precise medication in different populations, extending its use to infants as young as 6 months old. It provides a new domestic option for the standardized treatment of obstructive airway diseases caused by conditions such as bronchial asthma, acute/chronic bronchitis, and emphysema.

Pain Points in Pediatric Medication: "Effective" is Not Enough, It Must Be "Easy to Use and Adhere to".
Unlike adults, children, especially infants and toddlers, often face multiple practical challenges during medication: fear of injections, refusal to take medicine, fragile gastrointestinal function, and an underdeveloped respiratory system can all directly affect medication adherence and treatment continuity. The emergence of the Transdermal Drug Delivery System (TDDS) has provided a new approach to treating respiratory diseases in children. As the third major drug delivery route after oral administration and injection, transdermal patches allow drugs to be absorbed through the skin into the bloodstream. They offer multiple advantages, including once-daily application, long-acting sustained release, stable plasma concentration, fewer adverse reactions, and avoidance of the hepatic first-pass effect and gastrointestinal inactivation. They also support parent-administered dosing and can be discontinued at any time, significantly lowering the barrier to medication.
Data show that about one-third of children exhibit clinical signs of wheezing before the age of 3, and by age 6, nearly 50% of children have experienced at least one episode of wheezing[1]. Wheezing is one of the most common respiratory symptoms in children, with major causes including asthma, respiratory infectious diseases, abnormal development of the trachea and bronchi, or foreign bodies[2].
The "Guidelines for Rational Drug Use in Children with Wheezing Disorders"[2] point out that β2-agonists are commonly used bronchodilators for children with wheezing disorders. They have become a core therapeutic drug by relieving airway spasms and reducing airway mucosal hyperaemia and edema. Depending on their duration of action, they can be divided into short-acting β2-agonists (SABA) and long-acting β2-agonists (LABA).
As a "wearable LABA preparation", the tulobuterol patches are particularly suitable for pediatric patients who have difficulty with inhalation or have low adherence, and it is recommended by multiple authoritative guidelines and consensuses both domestically and internationally[3-5].
From Structure to Process: Designed to "Stay On and Deliver Stable Efficacy"

Deruituo® is not a simple "dose reduction" but involves process optimization at the formulation level. The product features a five-layer structure, including a backing layer, an adhesive layer, a drug layer, a rate-controlling membrane, and a protective layer, balancing both application stability and sustained-release performance. In terms of core technology, Deruituo® has secured two product patents:
● Introduction of a special compound into the adhesive layer to effectively enhance the stability of tulobuterol and inhibit drug crystal precipitation, solving the problems of decreased drug concentration and fluctuating efficacy caused by crystallization;
● Optimization of the glass transition temperature of the matrix material, allowing the patch to maintain good elasticity and adhesion at skin surface temperature, making it less likely to fall off during prolonged application and more suitable for children's activities.

Leveraging the technological advantages of the tulobuterol patches' long-acting sustained release, applying it to the chest, back, or upper arm before bedtime can effectively cover the periods of frequent attacks at night and in the early morning, improving the "morning dip" phenomenon and the patient's sleep quality. Many asthma attacks are thought to be caused by the "morning dip".
Respiratory function peaks at 4 p.m. and drops to its lowest point at 4 a.m., the so-called "morning dip". This time period is inconvenient for patient treatment and medication[6]. Patients with acute and chronic bronchitis, Chronic Obstructive Pulmonary Disease (COPD), pediatric wheezing diseases, and other conditions all experience problems such as worsening cough and wheezing in the morning, difficulty maintaining continuous sleep, and poor sleep quality.
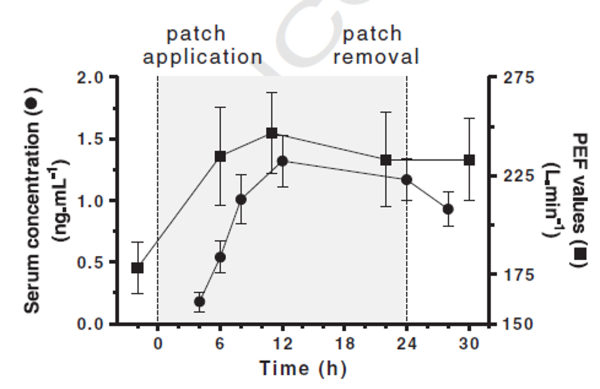
A pharmacokinetic study evaluating the use of the tulobuterol patches in children with moderate to severe asthma showed that the plasma concentration gradually increased within 4 hours of application and peaked at 12 hours.Meanwhile, the peak expiratory flow (PEF) of the children significantly improved after patch treatment and remained superior to the control group even after 22 hours[7]. In terms of adherence, a survey involving patients with asthma/COPD and parents of children with asthma showed that over 80% of patch users chose to "use as prescribed"[8].
Deeply cultivating the topical patch sector and adhering to the principle of "Good Patches, Made by Tide".
As a leading enterprise that has been deeply involved in the field of transdermal preparations for over 20 years, Beijing Tide Pharmaceutical has established a large-scale production capacity of 330 million patches per year. It adheres to the quality control standard of being "on par with or better than the originator" throughout the entire chain, ensuring stable product supply and consistent efficacy.
In 2010, Flurbiprofen Cataplasms (Debaian®) was approved for marketing, with annual sales exceeding RMB 3 billion. In 2018, the launch of Lidocaine Cataplasms (Debaining®) filled the gap in topical treatment for post-herpetic neuralgia. In 2025, a new indication was added for the treatment of diabetic peripheral neuropathic pain (DPNP), making it the world's first and currently only topical analgesic patch approved for this indication.
In the past two years, a series of patch products have been launched intensively, including the Rivastigmine Transdermal Patch (Suleda®) for Alzheimer's disease, the Tulobuterol Patches (Deruituo®) which ushers in a "patch era" for respiratory disease management, and the Rotigotine Patch (Luofeiding®) for Parkinson's disease, forming a product ecosystem covering multiple therapeutic areas such as pain management, central nervous system, and metabolism.
In the next 5 years, Beijing Tide Pharmaceutical plans to continue launching more than 10 transdermal drug delivery products, continuously strengthening the technological depth and clinical value of domestic patches. Currently, innovative patch products like Deruituo® are fully available on JD Health and in national chain pharmacies. Sino Biopharm will also work with partners nationwide to popularize related disease management and patch application methods in various forms, helping patients gain more convenient access to this innovative therapy.
References:
[1] Pediatric Respiratory Group, Pediatricians Branch of Chinese Medical Doctor Association; Editorial Board of Chinese Journal of Pediatrics; Respiratory Professional Committee of Futang Research Center of Pediatric Development. Clinical practice expert consensus on differential diagnosis and initial management of wheezing in children under 6 years of age [J]. Chinese Journal of Pediatrics, 2023, 61(4):301-309.
[2] Expert Committee on Pediatric Medication of the National Health and Family Planning Commission; Pediatric Respiratory Group, Pediatrics Branch of the Chinese Medical Association; Pediatric Respiratory Professional Committee, Pediatricians Branch of the Chinese Medical Doctor Association, et al. Guidelines for Rational Drug Use in Children with Wheezing Disorders [J]. Chinese Journal of Applied Clinical Pediatrics, 2018, 33(19):1460-1472.
[3] Nakamura Y, et al. Japanese guidelines for adult asthma 2020[J].Allergol Int.2020 Oct;69(4):519-548.
[4] Arakawa H, et al. Japanese guidelines for allergic rhinitis 2020[J].Allergol Int.2020 Jul;69 (3) :314-330.
[5] Global Initiative for Asthma (GINA).Global strategy for asthma management and prevention, updated 2023.
[6] TURNER-WARGARET M.On observing patterns of airflow obstruction in chronic asthma[J].Br J Dis Chest,1977,71(2): 73-86.
[7] Delgado-Charro MB, et al.Effective use of transdermal drug delivery in children.Adv Drug Deliv Rev. 2014 Jun:73:63-82.
[8] Tamura G, Ohta K. Adherence to treatment by patients with asthma or COPD: comparison between inhaled drugs and transdermal patch [J].Respir Med, 2007, 101(9): 1895-1902.
Disclaimer:
1. This press release is intended to facilitate the communication and exchange of medical information and is for reference by healthcare professionals only. It is not for advertising purposes.
2. The company does not recommend any drugs and/or indications.
3. The information contained in this press release is for reference only and cannot replace professional medical guidance in any way, nor should it be considered as a diagnosis or treatment recommendation. If you wish to understand specific information about disease diagnosis and treatment, please follow the advice or guidance of a doctor or other healthcare professional.